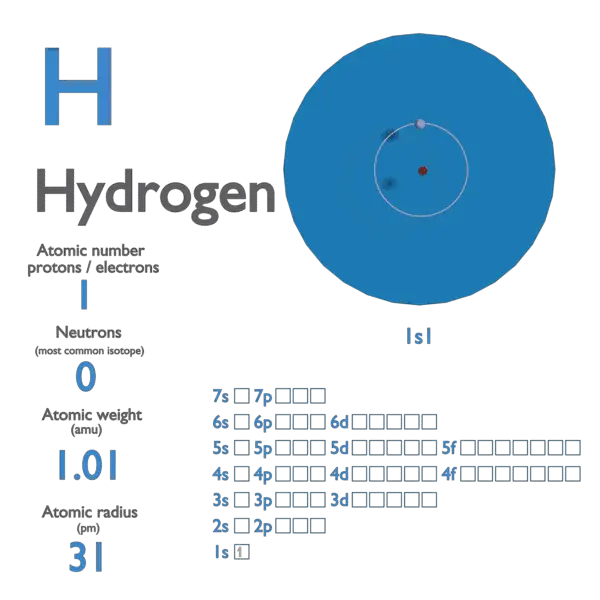
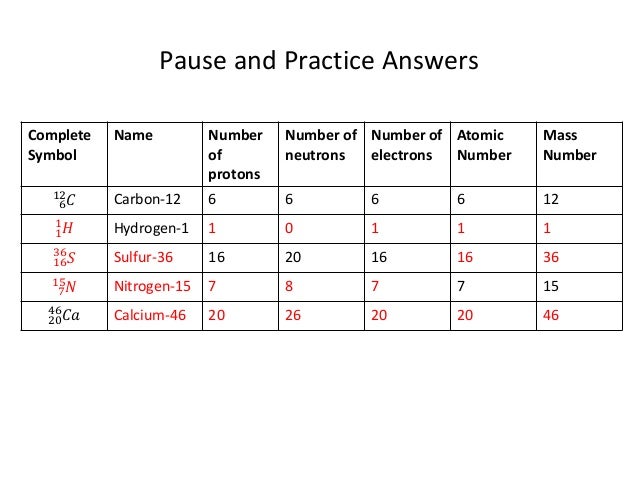
The atomic number of an element never changes, meaning that the number of protons in the nucleus of every atom in an element is always the same. Oxygen atoms contain 8 protons and have an atomic number of 8. All carbon atoms, and only carbon atoms, contain six protons and have an atomic number of 6. For example, all hydrogen atoms, and only hydrogen atoms, contain one proton and have an atomic number of 1. In other words, each element has a unique number that identifies how many protons are in one atom of that element. The number of protons in the nucleus of an atom determines an element's atomic number.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
